The impact of biofouling on surface membrane properties in MBRs

Youngpil Chuna,b, Dennis Mulcahya, Linda Zouc, In S. Kimd, Pierre Le-Clechb*
a Centre for Water Management and Reuse, School of Natural and Built Environments, University of South Australia, Mawson Lakes, Australia
b UNESCO Centre for Membrane Science and Technology, School of Chemical Engineering, University of New South Wales, Sydney, Australia, email: [email protected](*P. Le-Clech)
c Department of Chemical and Environmental Engineering, Masdar Institute of Science and Technology, Abu Dhabi, United Arab Emirates
d Global Desalination Research Center (GDRC), School of Environmental Science & Engineering, Gwangju Institute of Science and Technology (GIST), Gwangju, Republic of Korea
1. Biofouling and its analysis
As with other membrane separation processes, MBR membranes are prone to fouling generally and biofouling in particular by organic matter originating from the microbial cells. These biofoulants vary in concentration with the activated sludge characteristics, such as the mixed liquor suspended solids (MLSS) concentration and solids retention time (SRT) (and so the food/microorganism (F/M) ratio), as well as the feedwater chemistry. Membrane biofouling is also thought to be affected by the membrane surface properties, such as the charge (or zeta potential) and surface roughness. The surface charge in turn depends on the surface chemistry, and the presence of ionisable functional groups such as R-COOH, R-NH3+ and R-SO3H, or adsorbed anions, polyelectrolytes, ionic surfactants, and charged macromolecules from the solution. The charge also changes with pH and ionic strength, which means that the membrane–foulant interaction (and specifically the adhesion of foulants to the membrane) is similarly influenced by these solution conditions.
A study of the adhesion of biofoulants to membrane surfaces has been conducted with reference to the electrostatic and hydrophobic interactions. The aim was to try and determine which system properties are the most important in determining fouling. Two different hollow fibre (HF) polyvinylidene difluoride (PVDF) membranes, along with a cellulose ultrafiltration (UF) disc filter (Table 1), were used under different filtration modes. The membrane surface charge (zeta potential), and hydrophobicity (contact angle) were measured. The phospholipid content of the biofoulants was analysed to quantify the viable biomass in the membrane fouling layer. The extent to which these key properties impact on fouling was then appraised.
| Membrane | Chemistry | Properties | Filtration mode |
|---|---|---|---|
PVDF: polyvinylidene difluoride. 'Active filtration' includes physical cleaning cycle (rinsing with water) | |||
| UF | Regenerated cellulose | MWCO 30kDa, 150 mm diameter | Passive filtration |
| HF I | PVDF | Pore size 0.04 µm, 1.3 mm outer diameter | Passive filtration |
| HF II | PVDF | Pore size 0.04 µm, outer diameter 1.92 mm, inner diameter 0.9 mm | Active filtration |
The study was based on a small, 22L MBR pilot plant fitted with immersed HF membranes (Table 2) and fed with a sewage analogue (Table 3). The HF I membranes were immersed in the mixed liquor without filtration or cleaning, whereas the HF II membranes were operated under the conditions shown in Table 2. These two modes were referred to as ‘passive’ and ‘active’. A thoroughly washed membrane (WHF) was used as a control for representing the membrane surface properties of the unfouled material.
| Parameters | Value |
|---|---|
| Membrane surface area | 0.579 cm2 |
| Pressure | 9–15 kPa |
| Type of membrane | PVDF HF membrane, HF II |
| Influent flow | 55 mL/min |
| Normal operating pressure | 9–15 kPa (30 kPa max) |
| Solids retention time (SRT) | 20 d |
| pH | 5.6–7.6 |
| DO | 0.83–5.25 mg/L |
| MLSS | 4659–7530 mg/L |
| Chemicals | g/8 L–MilliQ water |
|---|---|
| Glucose | 126.4 |
| Sodium acetate | 101.6 |
| Ammonium chloride | 30.56 |
| Peptone | 28 |
| Meat extract | 17.2 |
| Monobase phosphate | 10.2 |
| Magnesium chloride | 8.8 |
| Iron(II) sulfate | 1.26 |
The contact angle measurements based on three test liquids of water (W), glycerol (G) and diiodomethane (D) were used to calculate the individual components of the membrane free surface energy γL. These components comprise the Lifshitz–Van der Waals, acidic, and basic components (γLLW, γL+ and γL− respectively) which combine to give γL (Table 4), a smaller contact angle indicating greater hydrophilicity and hence presumed to yield better adhesion.
| Liquid | γL | γLLW | γL+ | γL− |
|---|---|---|---|---|
| Water (W) | 72.8 | 21.8 | 25.5 | 25.5 |
| Glycerol (G) | 64 | 34 | 57.4 | 3.92 |
| Diiodomethane (D) | 50.8 | 50.8 | 0 | 0 |
2. Influence of membrane characteristics
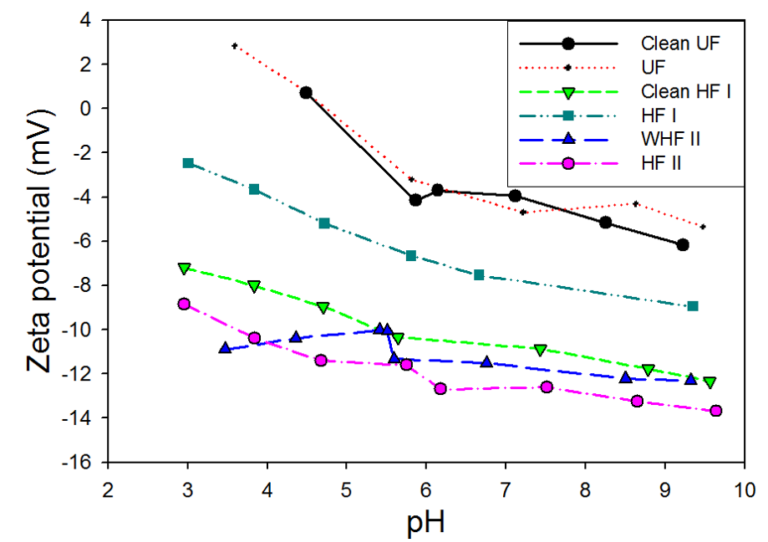
The rate of change of zeta potential with pH was roughly the same across all the materials other than the WHF one (Fig. 1). The zeta potential measurements showed the HF I and II membranes at pH values of 3−9 to be negatively charged over the entire pH range, whereas the more hydrophilic UF membrane appeared to have an isoelectric point at pH ~4.7. HF II gave more negative values than the other membranes samples, the negativity decreasing from HF II to HF I to UF. Whereas there was little apparent impact of biofouling on the UF and HF II membranes, the clean and fouled trends being essentially the same, the fouling had a marked impact on the zeta potential trend of the HF I membrane. This membrane was less negatively charged when fouled, presumably due to the presence of adsorbed hydrophobic organic matter.
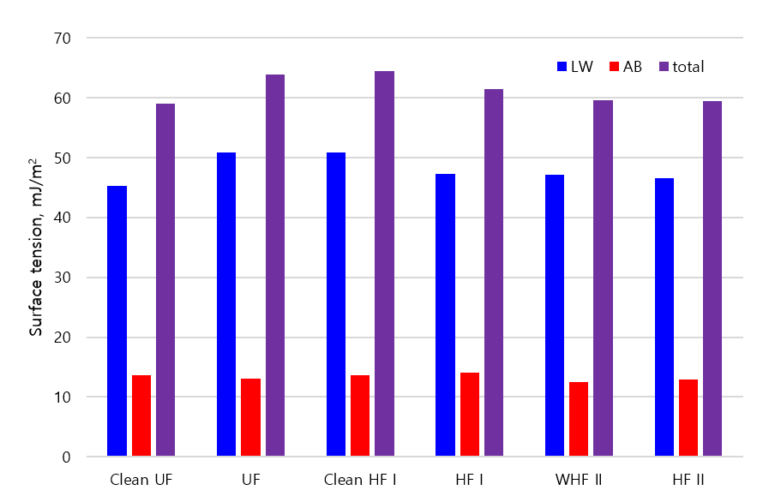
Contact angle measurements and corresponding calculated Lifshitz−Van der Waals (γSLW), Lewis acid–base (γSAB) and total (γtotal) surface tension values for the membrane samples (Fig. 2) differ only slightly across the different materials, despite the cellulosic material having been expected to be more hydrophilic. The effect of fouling on the membrane surface contact angles was also insignificant. Across all the materials tested there was just a slight decrease in the water contact angle resulting from fouling and thus no substantial change in the total free energy. The latter was low at 59−65 mJ/m2 and dominated by Lifshitz−Van der Waals intermolecular interactions. The acid–base forces varied by less than two mJ/m2 across all samples.
As with the zeta potential measurements, there was no significant difference between WHF II and HF II samples, i.e. the cleaned and fouled membrane material. This implies that, contrary to expectations, fouling did not significantly change the measurable surface charge and energy characteristics of the membrane. The total surface energy of the UF membrane increased only marginally, from 59 to 64 mJ/m2, whereas there was a similar decrease (from 65 to 61 mJ/m2) in this value for HF I. Biofouling thus could be said to have had a very slight negative impact on the nominally hydrophilic cellulosic UF material and a similar positive influence on the hydrophobic PVDF one. However, the impact in each case was very small.
3. Biofouling measurement based on phospholipids
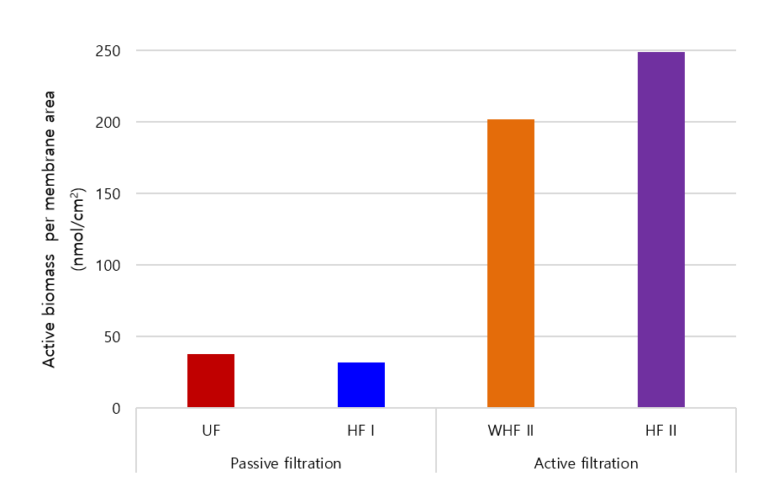
Phospholipids make up between 90 and 98% of bacterial cell membranes and have been widely adopted as a measure of viable biomass concentration. The results of the analysis (Fig. 3) indicated similar amounts of microbial biomass accumulated on the membrane for the different membrane materials (cellulose UF and PVDF HF) for similar passive filtration conditions. However, filtration mode (passive vs. active) and operation time (2 weeks vs. 3 years) impacted significantly on biofouling development, with active filtration over an extended time period producing seven-fold more deposited biomass. Active filtration produced permanent fouling, as indicated by the residual foulant surface concentration on the WHF membrane following extensive washing being only 19% less than that of the fouled membrane. However, such significant fouling did not appear to change the surface characteristics of the membrane.
4. Conclusions
The outcomes of the study suggest that the intrinsic surface properties of a membrane material are not significantly altered by fouling, regardless of whether the membrane is nominally hydrophobic or hydrophilic. The mode by which the membrane was fouled, i.e. through simple exposure of the material to the mixed liquor or by actual filtration, had a very significant impact on the amount of foulant deposited. Against this, the impact on the measurable material surface properties appeared to be negligible, other than in one case the zeta potential. The depositing of foulant on an MBR membrane may therefore be largely uninfluenced by the physicochemical properties of the membrane surface, and substantially be determined by the nature of the MBR operation and maintenance.