Ragging in MBRs − how do rags form?
Pompilia Buzatu1, Hazim Qiblawey1, Albert Odai1, and Simon Judd2
The authors are based in the following institutions:
1Qatar University, 2713 Doha, Qatar
2Cranfield University, Cranfield, Bedford MK43 0AL, UK
1. Introduction
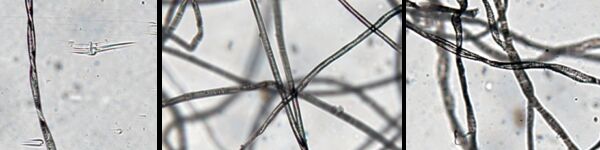
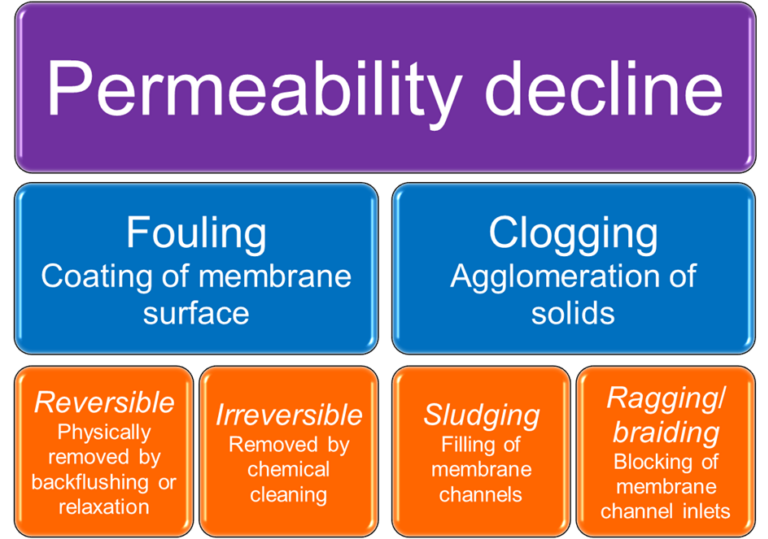
The phenomenon of ragging is familiar to many MBR operators. Ragging is a type of clogging (Fig. 1), where clogging relates to agglomeration of solids in the membrane tank. In the case of ragging, as opposed to sludging which relates to accumulation of solids within the membrane channels, the filament solids from textile materials join together to form long rags or braids (Fig. 2). These rags, which may contain filaments no more than a few mm in length, are mechanically stable in the mixed liquor and can block the inlet channels of the membrane module and/or wrap themselves around the infrastructure of the membrane tank – including the aerators.
Rags may form in both flat sheet and hollow fibre immersed MBRs (Fig. 2), but little is known of the factors contributing to their formation (or their 'ragging propensity'). Although there have been a small number of studies of the phenomena, such as at the University of Girona, there has been no attempt to try and replicate rag formation under controlled conditions. It has been suggested that the rags are formed by cotton products, and cotton wool specifically. However, this has never been categorically proven, and there is at least anecdotal evidence (from the University of Girona study) suggesting that other textile materials may contribute to rag formation.


2. The ragging test
A simple test has been developed at Qatar University to assess the ragging propensity of sample textile materials. To demonstrate the phenomenon, very small lengths of filaments are prepared, since one would expect the longer lengths to be removed by the fine screens upstream of the MBR.
The filaments are generated by cutting bundles of the textile material into lengths of less than 5 mm before they are mechanically disentangled to form single or very small discrete aggregates of fibres. They are subsequently carefully dispersed, slowly adding the fibres over a 3−4 hour period, in a 15 L volume of water at a low concentration of 5−10 mg/L (a total mass of ~100 mg fibres in total).
The filaments are then allowed to flocculate over a further 4−10 days under conditions of slow mechanical stirring (200−250 RPM). The stirrer speed selected is high enough to fully mix the entire volume of suspension but low enough to ensure rag formation is not disrupted or broken down by high shears.
Ragging in MBRs - rag dispersion
At the end of the test any self-supporting rags are extracted from the liquid, dried and their weight and length measured. The remaining suspension is filtered through a 0.45 μm-rated fibreglass filter and the collected material dried and weighed.
So far, the test has been conducted on fibres from three different sources (Fig. 3):
- commercial cotton wool balls normally used for make-up removal and readily available in pharmacies
- a sample retrieved from a rag formed in the membrane tank of a full scale MBR plant in the UK
- lint collected from a domestic tumble drier.
The rag from the MBR was washed, bleached and dried before use to remove all dissolved organic material (Stefanski et al, 2011). Both this sample and the cotton wool required preparation (cutting and mechanically separating) prior to the dispersal and ragging test, whereas the lint sample already comprised sufficiently short lengths of filament (<<5 mm on average) that were readily dispersed in water.

All ragging tests were replicated 5−6 times, and the starting materials also subject to optical microscopic analysis to attempt to identify the main textile constituent of each.
3. Results
3.1 Ragging trial
Data recorded across the study showed cotton wool to be more prone to rag formation than the other materials, forming longer rags in a shorter time period: 5 days, compared with 10 and 6 days for the MBR and lint samples respectively. Rag formation for the lint material was less consistent than for the other samples; only for one third of the tests conducted on lint were rags formed, whereas rag formation took place for all tests on the cotton wool and MBR sample filaments.
The characteristics of the rags differed between the different materials, with the cotton wool rags tending to be long and thin (Fig. 4a) compared with those from the MBR sample material (Fig. 4b) when both were extracted and suspended in air. Ragging was otherwise quantified in terms of (a) the average and cumulative length (for the formation of more than one rag) of the rags, and (b) the amount of material recovered as rags.


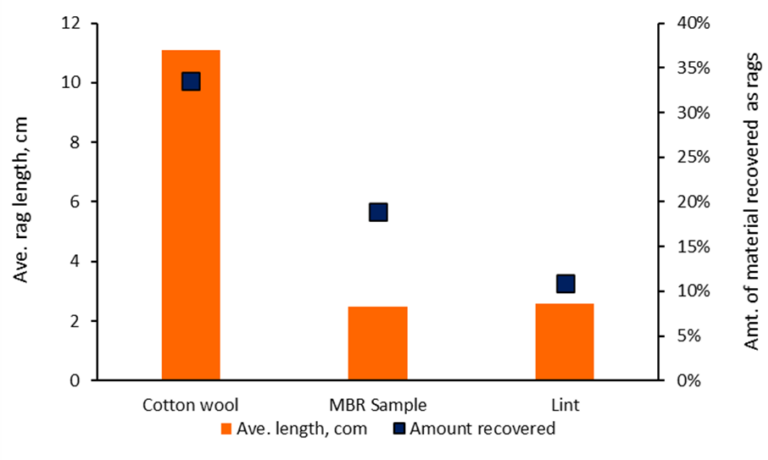
Data for rag length and amount of material recovered as rags (Fig. 5) indicated cotton wool to form longer rags than those formed by the MBR material or the lint. In the case of lint, the figure shown refers to those experiments where rags were formed (i.e. for 33% of all tests conducted on this material). The data for material recovery indicate that one third of the cotton wool added formed long rags, compared with just below a fifth for the MBR sample material and 11% for the lint.
The lint also formed spherical agglomerates around 2−3 mm in diameter (Fig. 6), making up around 13% of the added material. Their formation seemed to depend on the presence of some very small cotton-based agglomerates that acted as seeding material for the larger agglomerates. No such behaviour was observed in the other two sample materials, which comprised longer filaments.

3.2 MBR membrane fouling
A filtration test was performed with a set-up described elsewhere, using MBR mixed liquor generated from an industrial effluent. It was demonstrated that adding cotton wool filaments at a concentration of 5−10 mg/L to the sludge while it was being filtered resulted in rag formation in the mixed liquor (Fig. 7) within a time period of 12 hours. The addition of the filaments increased the transmembrane pressure (TMP) from 100 to 200 mbar within 1.5 hours.
The removal of the rags from the system did not result in the recovery of the TMP, suggesting that the membrane had become fouled as a result of the addition of the cotton wool. This was attributed to (a) the adherence of residual suspended filaments to the membrane surface and/or (b) the release of dissolved foulant material from the filaments.

Since dissolved organic foulants would be expected to be manifested as COD, COD measurement of both supernatant and filtrate generated from suspended cotton wool filaments in water was conducted. Results indicated elevated levels of COD in the supernatant (15 mg/L) compared with the filtrate (5 mg/L). This suggests, if only anecdotally, that cotton wool may contribute to membrane fouling, though clearly it would be expected for much of the dissolved organic foulant material to be leached from the filaments before reaching the wastewater treatment works.
3.3 Optical microscopic analyses
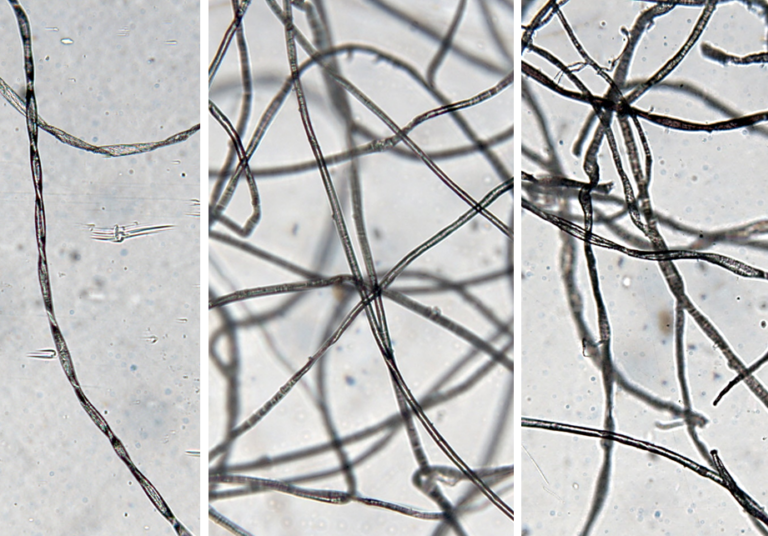
Cotton wool filament is characterised by a helical structure (Fig. 8a). Whilst this structure was also evident for the lint sample (Fig. 8c) it was less apparent in the MBR one (Fig. 8b). The MBR sample filaments instead seemed to mainly comprise synthetic fibres, and most probably nylon and acrylic. The fact that the filaments mainly comprised synthetic fibres warrants further investigation, since all other evidence points to cotton wool as being the main instigator of ragging. The increased propensity of the cotton wool fibres to form rags over the lint sample is most likely to be due to the comparative length of the original filaments; the lint filaments were substantially shorter in length.
4. Conclusions
It is evident from the simple tests performed that cotton wool has a high propensity for ragging, with around one third of the filaments added forming a single rag. This took place within the space of a few days and at very low filament solid concentrations (<10 mg/L). Moreover, the ragging phenomenon was repeated in a sample of mixed liquor.
There was some indication that ragging propensity may be linked to filament length. The use of lint as a starting material resulted in smaller agglomerates and a conversion of only around 11% of the starting material to rags. The only physical recognisable difference between the virgin cotton wool and the lint sample was the length of the filaments. Against this, a sample of rags taken from an MBR re-formed rags following dispersal, yet appeared to contain little cotton wool according to microscopic studies.
Lastly, it is possible that virgin cotton wool may leach a dissolved organic foulant material. However, this may not be relevant given that such leachates would be expected to have been largely removed by the time the cotton wool reaches the wastewater treatment works.
There is apparently still much to learn about the nature of ragging. Clearly, though, rags can be generated by very low concentrations of very short filaments. At 10 mg/L, the filaments make up around 0.1% of the total suspended solids in the membrane tank. Moreover, fibres of ~5 mm in length, as employed in this study, are likely to break through the fine screens generally employed in full-scale MBR installations (often rated at 2-3 mm).
The question that has to be answered is: 'What can be done?' The fact that long rags are formed in situ suggests that they should be readily removed by a relatively coarse screen. This would appear to advocate the installation of a coarse screen – perhaps rated at only 5 mm – on the return activated sludge (RAS) stream. Very few existing full-scale MBRs have RAS screening, but much could be learned from an examination of the screened material of those which do.